Resistance genes are not everlasting!
The resistance gene effect does not last forever. Although certain resistances remain effective for many years (for example, the resistance to phylloxera, introduced by American rootstock at the end of the 19th century), others lose their effectiveness more or less rapidly. One enlightening example is the resistance gene Rlm1 to oilseed rape stem canker (caused by the fungus Leptosphaeria maculans) that broke down in just three years in France (Rouxel et al., 2003). All plants and diseases together, half the proven cases of loss of effectiveness occurred in the first six years (McDonald and Linde, 2002).
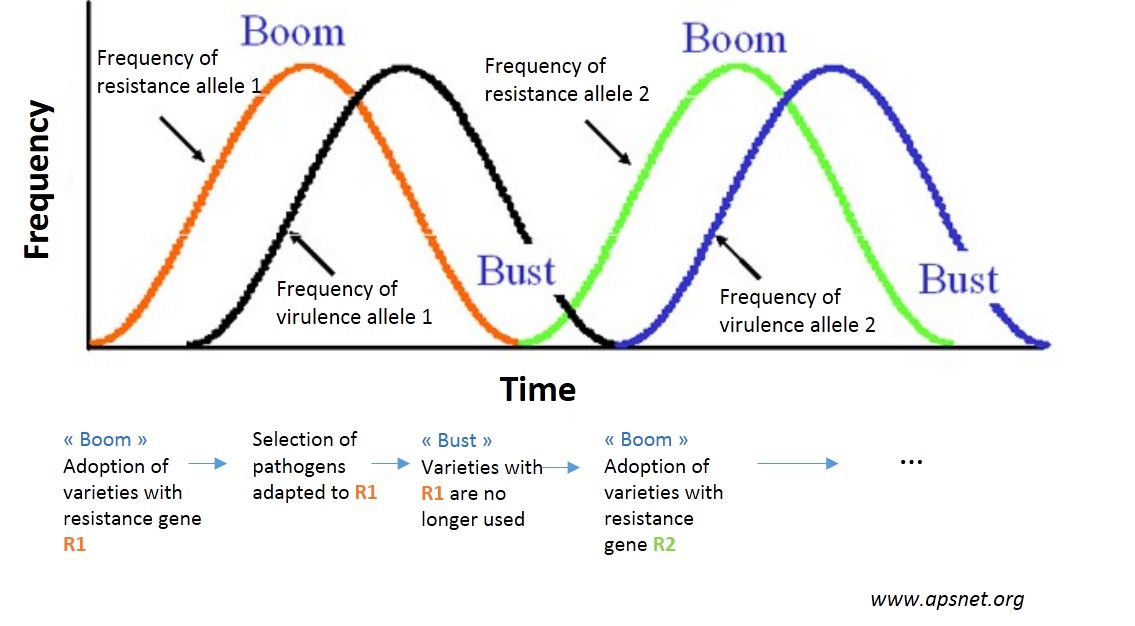
The reason for these losses in effectiveness? Just as bacteria adapt to antibiotics, so pests adapt to the resistant varieties according to the evolutionary biology1 mechanisms. These adaptations cause a gradual loss of effectiveness of resistant varieties. Agronomists talk about “breakdown” beyond a certain damage threshold. Other varieties with another resistance gene can then be introduced which will then be broken down and so on in a vicious cat and mouse cycle – the term used is boom and bust (see figure opposite).
These breakdowns can cause even more problems as frequently only a small number of resistances are available for a given pest (if indeed they exist at all!). Every breakdown therefore weakens the future of genetic control of plant diseases and pests…
Total resistance, partial resistance? Distinction is made between two major types of plant resistance. Total resistance: also known as qualitative resistance, it generates lack of disease. It is normally controlled by a major resistance gene, corresponding to an avirulence gene of the pathogen. Partial resistance: also known as quantitative resistance, it reduces the target disease. Unlike total resistance, it tends to be effective against all strains of a pathogen population.
The terms used when these resistances lose their effectiveness are breakdown for a qualitative resistance – the gene is now totally ineffective, it has broken down – and erosion for a quantitative resistance (Niks et al., 2015). Partial resistances are assumed to be more durable as less breeding pressure is applied to the pathogen. The resistance will last longer if a qualitative and quantitative resistance are combined in a single variety (Brun et al., 2010).
Vine resistance factors: a fragile heritage that must be protected
To date, fourteen mildew resistance factors (called Rpv) and eleven powdery mildew (called Run or Ren) have been identified in vines (Vitis spp). They form a fragile heritage, as their effectiveness and therefore their usefulness are subject to the risks of changes in pathogen populations, all the more so that:
- Vine powdery and downy mildew have huge evolutionary potential as they combine several characteristics that encourage them to adapt (production of a large quantity of spores, good dispersion capabilities, large population size in Europe because of the favourable climate conditions and virtually exclusive use of sensitive grape varieties, etc.). This huge evolutionary potential in vine downy and powdery mildew is illustrated by the speed with which these two pathogens have already responded to the pressure applied by the fungicides by developing resistances to almost all the molecules used in just a few years (Chen et al., 2007).
- In their original production area (North America), powdery and downy mildew are both capable of infecting an extensive rage of Vitis spp species, including some which are parents of recently-bred resistant grape varieties.
- Several proven cases of erosion of resistance genes have been documented for downy and powdery mildew: for example, a first c
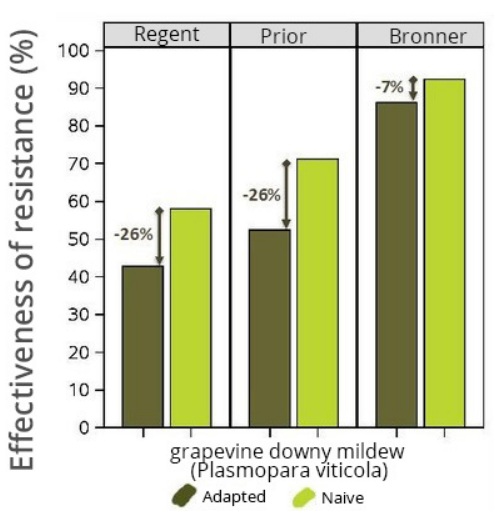 ase was described in 2010 on the Bianca variety in Europe and oidium was seen to have infected plants in North America that bore a resistance gene. Two studies conducted at INRA also revealed erosion in the resistance of the Regent, Prior and Bronner varieties deployed in Germany.
ase was described in 2010 on the Bianca variety in Europe and oidium was seen to have infected plants in North America that bore a resistance gene. Two studies conducted at INRA also revealed erosion in the resistance of the Regent, Prior and Bronner varieties deployed in Germany.
In such a context, the resistances introduced should ideally be managed durably, which involves a combination of strategies to reduce simultaneously 1) the development of short-term epidemics and 2) the probability of emergence of adapted strains in the long term.
What therefore are the strategies introduced for resistant grape varieties?
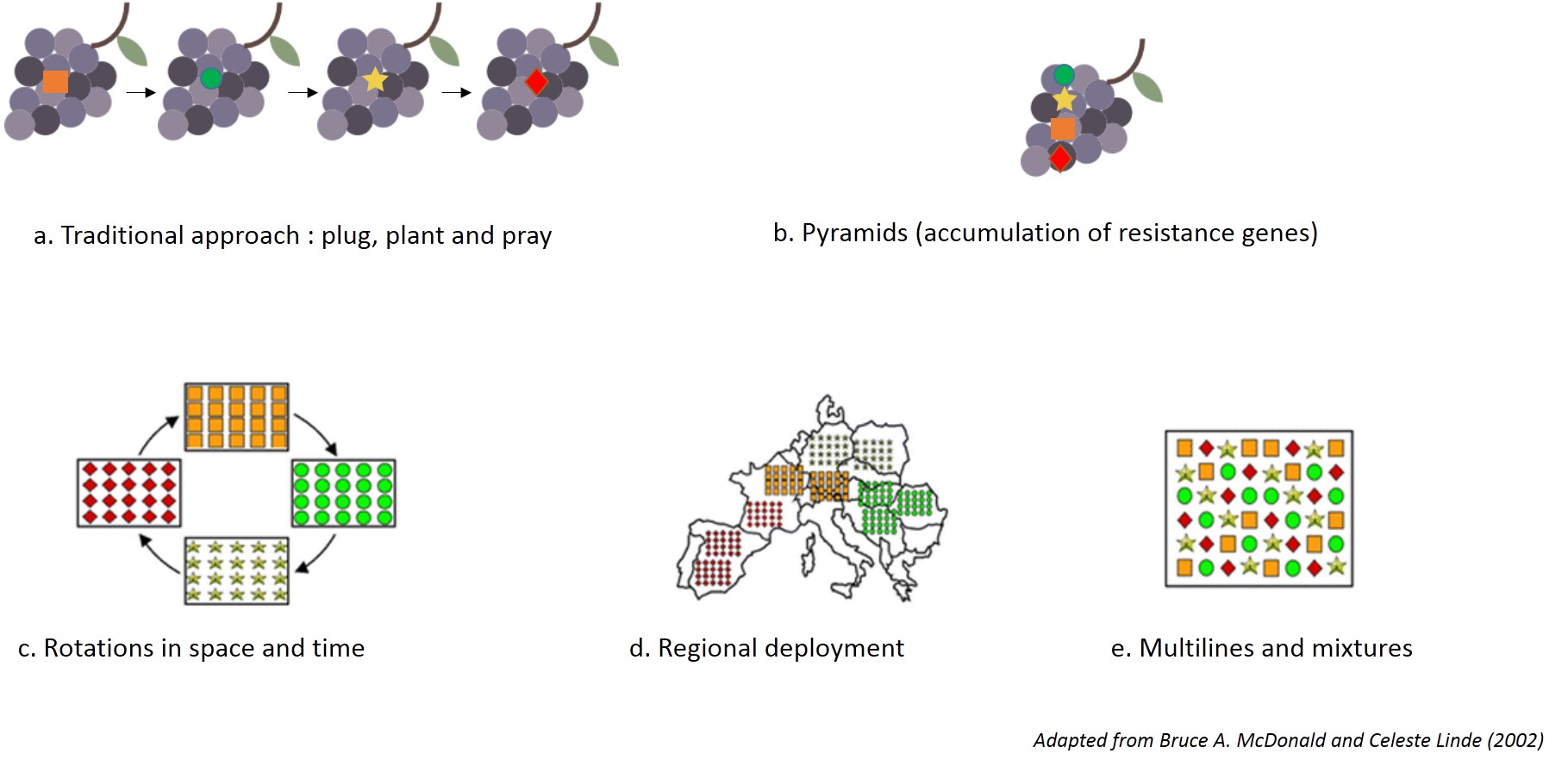
- 1. Pyramiding of resistance genes
Associating several resistance factors in a same variety, known as pyramiding, is likely to increase the durability of the resistance, by making breakdown more difficult. This is currently the strategy of the INRA varietal breeding program (Resdur) launched in 2000 and most European breeding institutes.
- 2. Adapted cultivation systems
Deploying resistant grape varieties in production conditions raises the question of their integration with the cultivation systems. Their use will in fact alter both the goals and the protection methods extensively. These cultivation systems should both supplement the partial effectiveness of resistance genes (especially for downy mildew) and guarantee their durability whilst controlling the diseases and pests that are not a genetic control target.
In practice, this will mean controlling the sizes of pathogen populations targeted by the resistance (downy and powdery mildew) and any other pathogens that may appear. A trial conducted by INRA Bordeaux since 2011 has seen the emergence of secondary diseases normally controlled by fungicides. The majority only cause minor damage (anthracnose, phylloxera), but others merit suitable protection strategies (for black rot and botrytis).
- 3. A national monitoring system: OSCAR!
The birth of OSCAR is going to secure the deployment of resistant grape varieties in France. Backed by a national monitoring network, the Observatory has many goals:
- Monitor the changes in populations of pathogens targeted by the resistance genes through regular collection of downy and powdery mildew isolates in the system;
- Detect the appearance of breakdowns and any appearance of new problems;
- Identify adapted cultivation systems
1 When the control method is applied systematically and is sustained over time – for example mass deployment of varieties with the same resistance gene – the resulting strong directional breeding lends itself to rapid adaptation by pathogens.